
In many ways, our generation is extremely fortunate when observed through the lens of history. Many of us will never know the pain of losing a sibling or child to polio, rheumatic fever, or diphtheria or the worry of minor cuts and scrapes becoming a lethal, untreatable infection. The advances of modern science and medicine have provided us with an arsenal of antibiotics to combat bacterial diseases, as well as effective vaccines that prevent many viral and bacterial infections from taking hold in the first place. However, our widespread abuse of antibiotics in both clinical and agricultural settings has led to an alarming increase in the amount of antibiotic resistant bacteria circulating in the environment and in our own bodies.
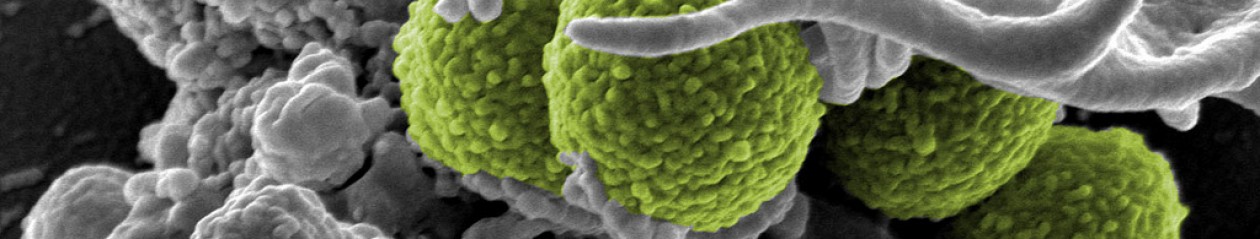
In the last few years many of us have heard of MRSA (methicillin-resistant staphlococcus aureus) in the news and its impact on health outcomes for those unfortunate enough to become infected. For those not familiar with the disease, MRSA is caused by the common bacteria Staphylococcus aureus, which is found all over the surface of our bodies and for the most part lives in relative harmony to us as a part of our normal flora. However, it is possible for Staph to become pathogenic given the proper circumstances such as a cut to the skin, foreign bodies such as splinters or catheters, immune compromise, or after treatment with antibiotics that disrupt the normal flora. In these unfortunate cases S. aureus can lead to a diverse array of clinical manifestations. In cases where the bacteria produces an exfoliative toxin that leads to Staphylococcal scalded skin syndrome (SSSS) where layers of the skin are shed in response to this toxin. Other clinical manifestations related to toxin production are toxic shock syndrome and food poisoning. Localized infections with this bacteria lead to impetigo, folliculitis, furuncles, carbuncles, and general wound infections. For more  unfortunate persons these infections can spread to the blood leading to a systemic infection and septicemia. In fact, invasive methicillin-resistant Staphylococcus aureus (MRSA) has become one of the leading causes of death in this country with approximately 6,000 MRSA related deaths in 2005 alone1. Even more frightening is that this bacteria is a common nosocomial infection- an infection acquired during a hospital stay. So a healthy person going in for a routine procedure runs the risk of acquiring a fatal infection.
unfortunate persons these infections can spread to the blood leading to a systemic infection and septicemia. In fact, invasive methicillin-resistant Staphylococcus aureus (MRSA) has become one of the leading causes of death in this country with approximately 6,000 MRSA related deaths in 2005 alone1. Even more frightening is that this bacteria is a common nosocomial infection- an infection acquired during a hospital stay. So a healthy person going in for a routine procedure runs the risk of acquiring a fatal infection.
So how has is come to pass that a part of our normal flora has become both pathogenic and highly resistant to antibiotics? A large part of this is due to our indiscriminant use of antibiotics for a multitude of applications that they are not necessary for. This may be surprising to some, but the bulk of antibiotics (70%) in this country are fed to our livestock in order to allow us to grow them in high population densities and increase their body mass before slaughter. They are not given therapeutically, but prophylactically in order to support the unnatural industrialized conditions in which they are produced. However, this high level of prolonged antibiotic use has flooded our environment with excess antibiotics and the microbes in these environments have responded in the most basic manner: survive however possible. This often means that bacteria develop resistance to these antibiotics. In fact, the FDA has known of this phenomenon since the 1970s but has yet to enact concrete bans on the non-therapeutic use of penicillin and tetracycline in animal feed.
This can be seen in humans as well when persons do not comply with the full antibiotic regimen and the remaining population of bacteria in that particular person is preferentially selected for antibiotic resistance. Additionally, physicians can and do prescribe antibiotics for conditions that do not require them. Viral infections are a case in point. A person will go to the doctor’s office complaining of flu-like symptoms and ask for antibiotics, even though these compounds have no effect against viruses or viral disease. In this case you once again knock-down those bacteria susceptible to the antibiotic and are left with a remaining population that is more resistant. Do this enough times and you will be out of treatment options.
What we are learning now is that antibiotics must be used with the utmost of care, lest resistance develops and we no longer have options for treatment. This evolutionary phenomenon is not limited to S. aureus as troubling antibiotic resistance has been observed in diverse bacterial species including but not limited to N. gonorrhea, E. coli, and M. tuberculosis. All of these pathogens can and do have profound impacts on public health.
In the case of Staphylococcus aureus we are almost out of defenses. Years ago it was noted that methicillin resistance (the common antibiotic used to treat Staph) was emerging in locations across the country and world. Now that this methicillin resistance has been firmly established newer, more powerful antibiotics must be used to treat infections. One of these last-line antibiotics is vancomycin and has been used successfully to treat MRSA infections. However, now it appears that Staph has once again outsmarted us and is gaining resistance to vancomycin, leading to a dangerous new subspecies: Vancomycin-resistant Staphylococcus aureus (VRSA)2. This is a strain that has now been isolated across the globe and has profound implications for the future of health care and the management of bacterial infections. What do we do when we cannot treat something as simple as an abscess? We are at the edge of an era in which our valuable tools against this unseen foe no longer work.
Steps are being taken to combat this threat. Recently the FDA updated recommendations that livestock should not be given antibiotics in their feed. However, this is just a recommendation and not a rule. Therefore, it is likely to be to totally ineffective as businesses notoriously do not adopt regulations that impact profit margins unless mandated to do so. At this point with the rapid emergence of multiple forms of antibiotic resistance bacteria we can no longer treat this as mere inconvenience, but a major hurdle in the way of increasing public health and safety. Steps need to be taken to stop the non-therapeutic use of antibiotics in our food supply as well as the off-target uses of antibiotics that is seen in clinical settings. Furthermore, much energy must be dedicated to the development of novel antibiotics that can target these resistance species so that unfortunate individuals who become infected have treatment options. By taking these steps we can work towards a future when routine procedures are safe and the infection of minor injuries does not turn into a death sentence. I’ll take that over a slightly fatter steak any day of the week.
Works referenced:
1. Klein, E., Smith, D.L. & Laxminarayan, R. Hospitalizations and deaths caused by methicillin-resistant Staphylococcus aureus, United States, 1999-2005. Emerging infectious diseases 13, 1840-6 (2007).
2. Kos, V.N. et al. Comparative Genomics of Vancomycin-Resistant Staphylococcus aureus Strains and Their Positions within the Clade Most Commonly Associated with Methicillin-Resistant S. aureus Hospital-Acquired Infection in the United States. mBio 3, (2012).
Reuter’s Article on recent ruling of FDA and antibiotics in animal feed: http://www.reuters.com/article/2012/06/05/us-fda-antibiotics-idUSBRE85417N20120605
Clinical manifestations of Staphylococcal disease found in: Medical microbiology / Patrick R. Murray, Ken S. Rosenthal, Michael A. Pfaller. -6th ed. Pgs 209-223
Perhaps vectored immunoprophylaxis could be used to protect against these infections by producing the correct monoclonal antibodies?
That’s a perfect use for the mature technology. It’s going to take a while before we can even think about using vectored immunoprophylaxis on a large scale in humans though. There’s a LOT of testing that needs to be done, but some of that work has already started. It’s a really exciting topic to follow.